What Is the Difference Between Peptides, SARMs, and Bioregulators?
In 2026, peptides, SARMs, and bioregulators are often grouped together online, but they are chemically, biologically, and legally distinct categories.
Understanding these differences is essential for researchers navigating compliance and regulatory risk.
- What Are Peptides?
Peptides are short chains of amino acids that function as signaling molecules in biological systems.
They may:
• Mimic hormones
• Influence cellular communication
• Act as growth factors
• Modulate metabolic pathways
Some peptides are FDA-approved drugs. Many are sold strictly for laboratory research use.
Examples of widely researched peptides include:
• BPC-157
• CJC-1295
• Ipamorelin
• Epitalon
Legal Status of Peptides (2026)
Peptides fall into three categories:
• FDA-approved pharmaceutical peptides
• Compounded peptides (regulated under 503A/503B)
• Research-use peptides (not approved for human consumption)
Regulatory reference:
Federal Food, Drug, and Cosmetic Act (FD&C Act) (view source)
For more on peptide legality, see our full guide: Peptides FDA Legal Status 2026
- What Are SARMs?
Selective Androgen Receptor Modulators (SARMs) are synthetic compounds designed to selectively bind to androgen receptors.
Unlike peptides, SARMs are small molecules — not amino acid chains.
Examples include:
• Ostarine
• Ligandrol
• RAD-140
Are SARMs Legal?
In 2026:
• SARMs are not FDA-approved dietary supplements.
• Many are classified as investigational new drugs.
• Some may fall under DEA scrutiny depending on classification.
• They are banned in professional sports by World Anti-Doping Agency (WADA).
FDA warnings on SARMs (view source)
SARMs carry higher enforcement visibility compared to most research peptides due to performance-enhancement marketing history.
- What Are Bioregulators?
Bioregulators are typically short peptides (often 2–4 amino acids long) derived from tissue extracts and studied primarily in Eastern European research traditions.
They are sometimes referred to as “cytomedins.”
Example:
• Epithalon
Bioregulators are theorized to:
• Influence gene expression
• Support tissue-specific cellular signaling
• Act via epigenetic pathways
Unlike SARMs, bioregulators are peptide-based.
Unlike most Western peptides, many originate from Russian research programs in the late 20th century.
Mechanism Differences
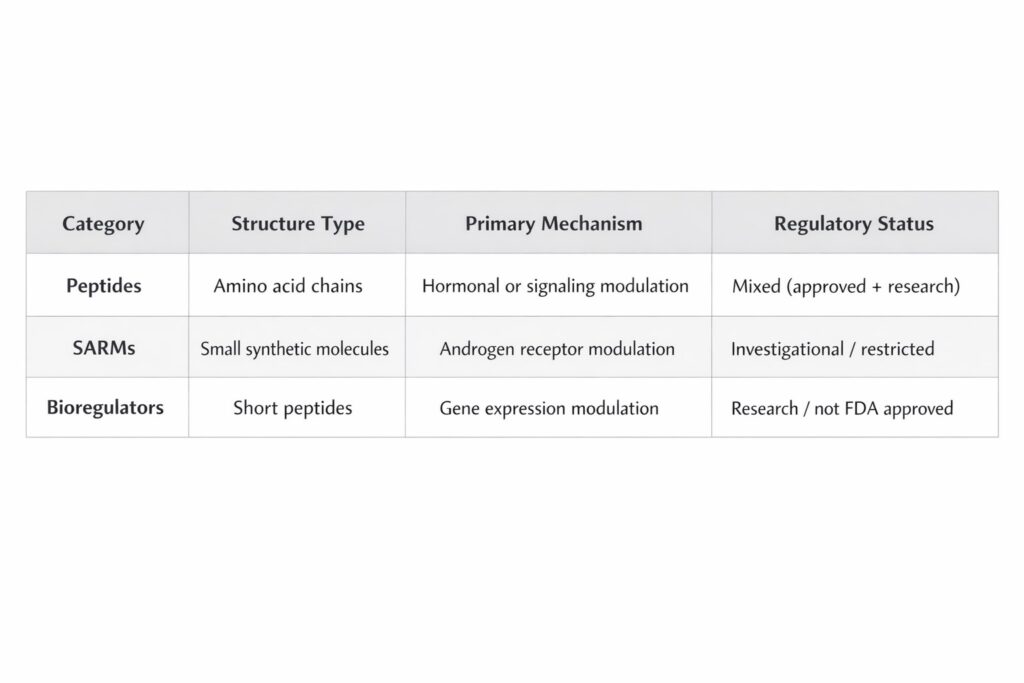
Peptides vs SARMs: Regulatory Risk Comparison
SARMs have historically drawn more FDA enforcement due to:
• Bodybuilding marketing
• Supplement mislabeling
• Hormonal manipulation claims
Peptides, when marketed strictly as research-use compounds without therapeutic claims, exist in a clearer compliance lane.
Bioregulators occupy a niche research category and are generally not classified as anabolic agents.
Which Category Is FDA Approved?
As of 2026:
• Some peptides are FDA-approved drugs.
• SARMs are not FDA-approved for general consumer use.
• Bioregulators are not FDA-approved pharmaceutical drugs in the U.S.
You can verify drug approvals in the FDA database (view source)
Clinical trial data is available via:
https://clinicaltrials.gov/
Why This Distinction Matters for Researchers
Blurring these categories creates:
• Regulatory confusion
• Compliance risk
• Marketing violations
• Platform deactivation risk
For research suppliers, clarity is protection.
At MainPeptides.com, compounds are:
• Sold strictly for laboratory research use
• Not intended for human consumption
• Not marketed with therapeutic or performance claims
Explore:
• Research Guides
• Peptide Storage Best Practices
• Partner Program
Frequently Asked Questions
Are SARMs considered steroids?
No. SARMs are not anabolic steroids, but they act on androgen receptors and are often regulated more aggressively than peptides.
Are bioregulators the same as peptides?
Bioregulators are a subset of short peptides, often studied for tissue-specific signaling.
Which is more regulated: SARMs or peptides?
Generally, SARMs face higher enforcement scrutiny due to misuse in the supplement market.
The Bottom Line
Peptides, SARMs, and bioregulators are not interchangeable.
They differ in:
• Chemical structure
• Mechanism of action
• Regulatory pathway
• Enforcement visibility
Understanding these differences is essential for compliant research operations in 2026.

